Pre-analytical blood collection tubes specifically for liquid biopsy, validated for collection of whole blood and stabilization of cfDNA and ctDNA for up to 21 days.
Longer stability Test
Stabilizes whole blood for up to 21 days at a wide range of storage temperatures (4°C – 37°C) taking away the worry of transport delays.
Easy to use
Plastic evacuated tube compatible with standard draw equipment and disposables so there is no need to change anything in your workflow.
Fully validated
Validated for use in non-invasive applications, such as NIPT and liquid biopsy; and for multiple techniques like qPCR and NGS.
Seamless workflow
Barcoded labels allow seamless workflow integration with your existing automation and laboratory management systems.
The importance of blood collection methods in cfDNA applications
The introduction of non-invasive methods like liquid biopsy that use cell-free DNA (cfDNA) for molecular diagnostics has been of growing interest in oncology and prenatal studies. The analysis of cfDNA in a biological fluid, usually blood, can provide valuable genomic information for healthcare professionals without the need for tissue biopsies or amniocentesis.
Whilst taking a blood sample is a simple procedure, if blood plasma is not preserved or isolated within 24 hours of blood draw, the degradation of nucleated white blood cells will cause the release of intracellular genomic DNA (gDNA) into the plasma. This contaminates the cell-free DNA fraction affecting the reliability and validity of data. This time constraint means that any delays in transport of samples from collection site to analysis location can negatively impact on results.
Nonacus Cell3™ Preserver tubes are an easy-to-use direct-draw blood collection method that maintains white blood cell integrity for up to 21 days. They provide an alternative to EDTA tubes that help ensure contamination-free cell-free DNA for applications like liquid biopsy and NIPT.
Designed for Direct-Draw Blood Collection
Cell3™ Preserver tubes consist of sterile, evacuated, polyethylene terephthalate tubes that are designed for direct-draw blood collection. They contain an additive at the correct volume to simultaneously stabilize blood cells and impede coagulation at the time of collection. The stabilizer acts by preserving the nucleated cells (white blood cells) in an intact state until processing and analysis can be performed.
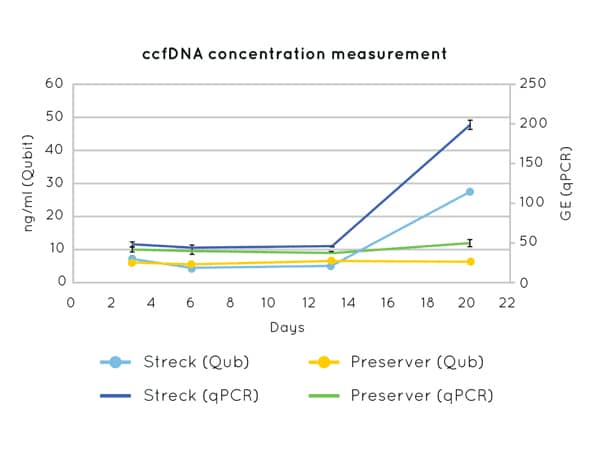
Figure 1. Streck tubes show an apparent increase in cfDNA concentration from day 13 due to contaminating gDNA released by the lysis of white blood cells.
Superior blood cell stability for cfDNA longevity
Cell3™ Preserver has superior blood cell stability compared with the market leader allowing blood samples to be stored for up to 21 days after draw at an ambient temperature before processing. This minimises the adverse effects of time, storage, and transport conditions on sample integrity and cfDNA extraction yield.
A cost-effective preserver validated for NIPT and liquid biopsy testing
Cell3™ Preserver has been validated for NIPT and liquid biopsy applications alongside the market leading cell stabilization tube and offers an alternative to standard EDTA blood collection tubes that ensures robustness of genetic testing with cfDNA.
Figure 1. DNA concentration of PCR-free libraries prepared from ccfDNA for whole genome sequencing on NextSeq500 (Illumina).
Blood samples from 5 patients were collected into both Cell3™ Preserver and Streck tubes for testing. CcfDNA was extracted and analyzed with the Serenity test for NIPT of fetal aneuploidies.
Figure 2. Fetal fraction observed
Blood samples from 5 patients were collected into both Cell3™ Preserver and Streck tubes for testing. CcfDNA was extracted and analyzed with the Serenity test for NIPT of fetal aneuploidies.

Figure 3. Liquid biopsy test: Two identified pathogenic variants were found present in the FFPE and cfDNA samples, but not in the germline dDNA.
A blood sample from a patient with metastatic breast cancer was collected in Cell3™ Preserver and Streck tubes post chemotherapy. CcfDNA was extracted and tested alongside gDNA obtained from leukocytes and a FFPE biopsy sample of the primary tumor. Targeted resequencing across a panel of genes commonly associated with cancer was performed at high depth Illumina). Bioinformatics analysis was conducted to annotate variants and identify causative actionable mutations.
Designed to support streamlined workflows
The Cell3™ Preserver tube has been designed for sample collection and shipment from around the world helping centralized lab facilities streamline their workflows. A sample barcode is included to allow LIMS compatibility, sample tracking and to assist high throughput clinical and translational labs.

 Forgalmazott termékeink gyártói - keressen gyártó szerint a logóra kattintva
Forgalmazott termékeink gyártói - keressen gyártó szerint a logóra kattintva